News outlet VICE, known for its investigative journalism, has just debuted a three part documentary that takes a look at Truvada and its use … [Read more...] about VICE Documentary Explores What ‘The Truvada Revolution’ Means For The Fight Against HIV: VIDEO
Truvada
New Study Sheds Light On Truvada’s Ability To Prevent HIV Infection After One Week of Dosing
A new study into Truvada’s efficacy as a form of pre-exposure prophylaxis (PrEP) has found that the drug is most effective at preventing HIV … [Read more...] about New Study Sheds Light On Truvada’s Ability To Prevent HIV Infection After One Week of Dosing
New PrEP Studies Show 86% Reduction In HIV Transmission Rates, Even As ‘On-Demand’ Treatment
Truvada taken as an HIV preventative is shown to be an incredibly effective way of stopping the spread of HIV. In 2014 the iPrEx OLE study showed that … [Read more...] about New PrEP Studies Show 86% Reduction In HIV Transmission Rates, Even As ‘On-Demand’ Treatment
PrEP Is Not The Problem, Perfection Is
The Interplay is a special biweekly series exploring the intersections of sex, pop culture, and current events. BY CHARLES … [Read more...] about PrEP Is Not The Problem, Perfection Is
Research on Injectable, Longer-Lasting PrEP Moving Forward
Studies focusing on injectible pre-exposure prophylaxis as a means of preventing the contraction of HIV is moving forward as researchers have seen … [Read more...] about Research on Injectable, Longer-Lasting PrEP Moving Forward
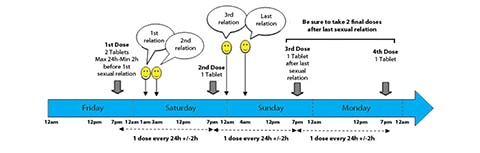
Major HIV News: ‘On-Demand’ PrEP Study Appears Successful
In major news in the fight against HIV, a second European study has concluded the "randomized" phase of its examination of the efficacy … [Read more...] about Major HIV News: ‘On-Demand’ PrEP Study Appears Successful
Major HIV News: ‘On-Demand’ PrEP Study Appears Successful
In major news in the fight against HIV, a second European study has concluded the "randomized" phase of its examination of the efficacy … [Read more...] about Major HIV News: ‘On-Demand’ PrEP Study Appears Successful
HRC Endorses PrEP, Truvada: READ
On Saturday, the Human Rights Campaign endorsed Pre-Exposure Prophylaxis (PrEP) - an HIV prevention strategy that reduces the risk of infection by the … [Read more...] about HRC Endorses PrEP, Truvada: READ
SF Supervisor Scott Wiener Comes Out of the PrEP Closet: ‘Each Morning, I Take a Pill Called Truvada’
San Francisco Supervisor Scott Wiener decided to "come out of the PrEP closet" this week in order to educate the public and destigmatize … [Read more...] about SF Supervisor Scott Wiener Comes Out of the PrEP Closet: ‘Each Morning, I Take a Pill Called Truvada’
Andrew Sullivan and Rich Juzwiak Continue Their Discussion on Gay Sex, Truvada: AUDIO
Andrew Sullivan sat down with Gawker's Rich Juzwiak to continue their discussion of Pre-Exposure Prophylaxis (PrEP) and to dissect the thought … [Read more...] about Andrew Sullivan and Rich Juzwiak Continue Their Discussion on Gay Sex, Truvada: AUDIO